
NEW DELHI: Twenty-one hospitals across the country have been granted permission to conduct stage 2 clinical trials to assess the safety and efficacy of convalescent plasma therapy, which has shown some promise in treating moderate to severe COVID-19 cases across the world.
These include five hospitals in Maharashtra; four in Gujarat, two each in Rajasthan, Tamil Nadu, Madhya Pradesh and Uttar Pradesh, and one each in Punjab, Karnataka, Telangana and Chandigarh.
The trials, to be carried out on 452 patients, however, will not have a placebo arm — which means there cannot be a comparison made between two groups of patients for the efficacy of therapy.
“Project PLACID — phase-2 open-label, randomized controlled trial of convalescent plasma by ICMR has received the approval of COVID-19 national ethics committee,” said Lav Agarwal, joint secretary, the health ministry in a briefing on COVID-19 status in the country on Friday.
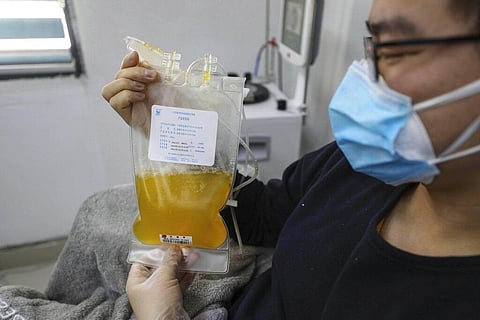
In convalescent plasma therapy, a dose of anti-body-containing plasma obtained from the blood of recovered individuals is transfused to persons with the disease to treat it.
It’s an experimental therapy, going back a hundred years, has found useful in many diseases and conditions.
It was tested in case of viral outbreaks during the treatment of the infamous 1918 Spanish flu as well as, more recently, during the 2009 H1N1 influenza, SARS and MERS virus outbreaks.
The Central Drug Controller of India had last month finalised protocol for the therapy saying that those recovered can donate blood after 28 days of recovery and cannot be asked to donate more than 1,000 ml of blood within a month.